We specialize in tick-borne diseases. It's the only thing we do.
HOW IGENEX BEGAN

Scientists from the ImmunoBlot department.
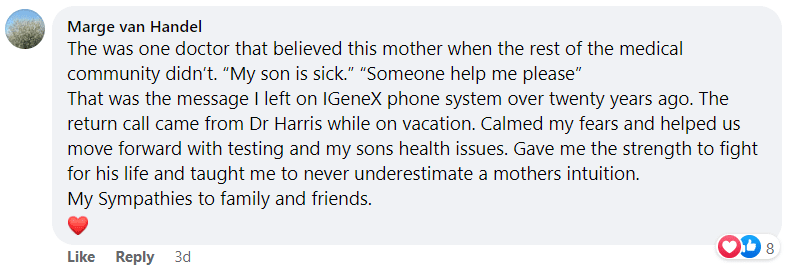
It all began after 3M sold its Diagnostics Division in 1991 and the leading pioneer in Lyme disease, Dr. Nick Harris, formed IGeneX with a small team of seven people. Dr. Harris realized early on that there was an urgent need for more sensitive Lyme tests. With his steadfast dedication to the scientific diagnosis of Lyme disease, he introduced groundbreaking laboratory testing to the scientific community with new diagnostic tools that have greatly improved the ability of physicians to diagnose these often debilitating diseases.
Dr. Harris was also instrumental in the formation of the International Lyme and Associated Diseases Society (ILADS). This organization is at the forefront of training physicians to recognize and treat tick-borne diseases and become Lyme literate physicians. His dedication ensured that future generations of physicians would receive the knowledge to provide the much-needed care for patients suffering from tick-borne diseases.
DR. JYOTSNA SHAH JOINS IGENEX IN 1997
Dr. Jyotsna Shah has over 40 years of research experience in the development of diagnostic tools for infectious diseases. She has authored multiple publications, and holds more than 20 patents.
When she joined IGeneX as a scientific researcher, it became clear to Dr. Shah that the commonly accepted testing criteria for Lyme disease were both too restrictive and incomplete. By working in close consultation with top scientists vested in Lyme research, physicians who were treating patients, as well as performing hands-on development in the lab, Dr. Shah and her team were able to develop their own in-house testing criteria.
The new criteria uses a wider spectrum of relevant proteins found throughout all the stages of infection, as well as from different parts of the world. This has led to the development of tests that find more true positives without sacrificing specificity.

Dr. Shah in the lab with Lab Director Iris Cruz.
INTO THE FUTURE

At IGeneX, it is our mission to continue to improve testing for tick-borne related diseases. As we have learned more about the tick-borne disease causing organisms, we have increased our menu of tests. We now offer testing for Lyme disease, Tick-Borne Relapsing Fever, Bartonella, Babesia, Rickettsia, Anaplasma, and Ehrlichia. The testing methods include ImmunoBlots, Western blots, PCR, FISH, IFA, and IgXSpot.
At IGeneX, we spend an industry leading excess of 15 to 20 percent of our net profits on research and development to continuously improve our diagnostic testing. We look for more and detect more without a corresponding loss in specificity, no matter when patients were exposed to the tick-borne pathogen.
IN LOVING MEMORY

Dr. Nick Harris
1942 - 2022
It is with deep sadness that we announce the passing of IGeneX Founder Dr. Nick Harris after a lengthy illness. Dr. Harris formed IGeneX in 1991 with a small team of seven people and was a great leader of the company for many years. He went on to develop groundbreaking laboratory testing for the Lyme disease community that has greatly improved the ability to diagnose this and related, often debilitating tick-borne diseases.
With his steadfast dedication to the scientific diagnosis of Lyme disease and his passion for educating the Lyme community, it was no surprise that he was also one of the founding members of International Lyme and Associated Diseases Society (ILADS). In addition, it was not unusual for Dr. Harris to spend hours on the phone providing guidance to patients and doctors. Nick touched the hearts of all who worked with him and will be missed by many.
He was surrounded by his loving family in his final days and passed comfortably and quietly. The world of medicine is better off because of Dr. Nick Harris. May he rest in peace.
We invite you to leave a comment on social media.

Dr. Nick Harris
1942 - 2022
It is with deep sadness that we announce the passing of IGeneX Founder Dr. Nick Harris after a lengthy illness. Dr. Harris formed IGeneX in 1991 with a small team of seven people and was a great leader of the company for many years. He went on to develop groundbreaking laboratory testing for the Lyme disease community that has greatly improved the ability to diagnose this and related, often debilitating tick-borne diseases.
With his steadfast dedication to the scientific diagnosis of Lyme disease and his passion for educating the Lyme community, it was no surprise that he was also one of the founding members of International Lyme and Associated Diseases Society (ILADS). In addition, it was not unusual for Dr. Harris to spend hours on the phone providing guidance to patients and doctors. Nick touched the hearts of all who worked with him and will be missed by many.
He was surrounded by his loving family in his final days and passed comfortably and quietly. The world of medicine is better off because of Dr. Nick Harris. May he rest in peace.